Basic Specifications
Sterilization
Ethylene Oxide Sterilization
Guarantee Period
Five Years
Logo Printing
Without Logo Printing
Product Description

- Made of non-toxic PVC, medical grade.
- Length: 40cm ± 2cm (Infant) / 120cm ± 2cm (Adult).
- Transparent design, with X-ray visualization available.
- Available Sizes: F4, F5, F6, F8, F10, F12, F14, F16, F18, F20, F22, F24.
- Supplied sterile in individual peelable polybag or blister pack.
- Sterilized by ethylene oxide (EO).
Manufacturing Capabilities
Factory Expertise
Our ISO13485 certified manufacturing facility specializes in tooling and molding for medical products. With over a decade of experience, we have exported thousands of molds to over 30 countries globally.
Our production expertise covers diverse fields including Infusion and Transfusion, Syringes, Hemodialysis, Respiratory care, surgical instruments, and Diagnostic equipment.
Clean Room Production
We offer Plastic Injection Molding within an ISO Class 8 clean room environment. Our OEM molding services are tailored for plastic parts of Class III medical devices, such as Catheter and Delivery System components.

Engineering & Tool Design
Design for manufacturability and efficient production is our primary objective. Utilizing advanced CAD/CAM/CAE systems (Pro-E, UG, AutoCAD, mold flow, power milling), we support various file formats:
- STEP, PRT, SLDPRT
- X_T, DXF, and more
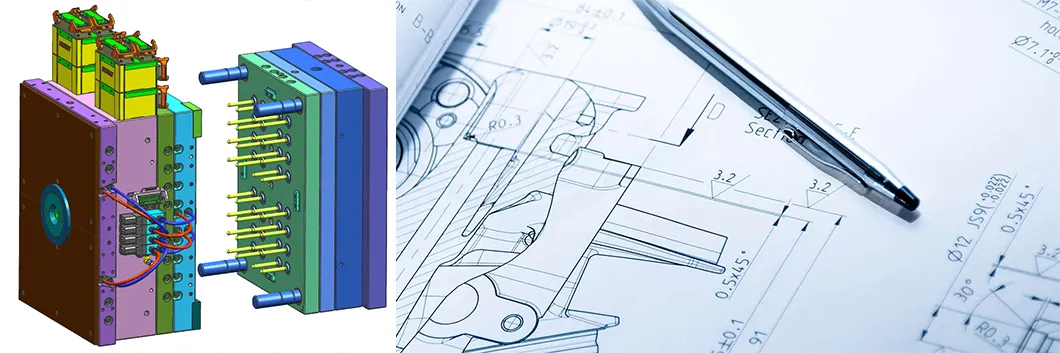
Our sophisticated design team ensures compliance with your production capacity requirements for various medical markets including IV Catheters, Syringes, and Diagnostic Devices.

Global Presence & Quality
International Exhibitions
We regularly attend international medical exhibitions such as CMEF, MEDICA, Arab Health, and FIME to stay at the forefront of medical technology.

Certifications

Frequently Asked Questions (FAQ)
Q1: What materials are used in the feeding tubes?
A: The tubes are manufactured using non-toxic, medical-grade PVC for patient safety and comfort.
Q2: How are the medical components sterilized?
A: All components are sterilized using Ethylene Oxide (EO) sterilization to ensure they are medical-grade and sterile.
Q3: Are the feeding tubes visible under X-ray?
A: Yes, the tubes are designed with X-ray transparency for easy positioning and verification during medical procedures.
Q4: What sizes are available for the sterile feeding tubes?
A: We offer a comprehensive range of sizes from F4 to F24 to accommodate both infant and adult patients.
Q5: What is the shelf life of the sterile products?
A: Our sterile medical components come with a quality guarantee period of five years.
Q6: Do you offer custom molding for medical parts?
A: Yes, we provide OEM injection molding services in an ISO Class 8 clean room environment for various medical device components.






















